Real-World Evidence Portfolio
Real-World Data (RWD) is patient data routinely collected in electronic health records (EHRs), claims data, and registries that can provide valuable information about the real-world performance of cancer therapies and diagnostics. Unlike in traditional clinical trial settings where data are collected at pre-established timepoints and reported uniformly for trial participants, there is typically a lot of variation in the way RWD are reported within and across sources. Inconsistent definitions and data missingness in RWD present challenges to using these data sources to investigate treatment effectiveness, such as how a therapy impacts survival or can be used in certain patient populations. Strategies and methodologies for mitigating these factors and aligning RWD are needed to fully realize its potential.
Since 2017, Friends has facilitated collaboration among RWD vendors, pharmaceutical companies, government officials, and academics to advance the understanding and applications of RWD and real-world evidence (RWE) in oncology. These multistakeholder partnerships support alignment on best practices, provide a venue to develop and test methodologies for aligning and analyzing RWD, and help identify opportunities to proactively strategize on future use of RWD/E in oncology:
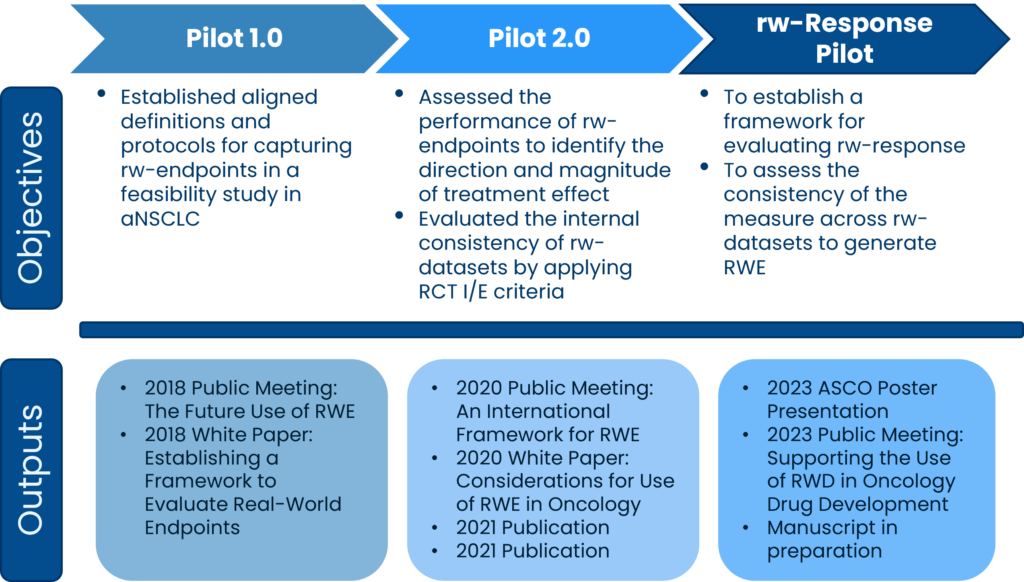
RWE Pilot 1.0 created a framework for aligning data sources and showed that implementing a harmonized protocol and set of definitions for necessary data elements supports identification of similar patient populations across data sources and extraction of several real-world endpoints.
RWE Pilot 2.0 applied the framework developed in Pilot 1.0 and showed that RWD can generate similar results to clinical trials when measuring the treatment effect of chemotherapy compared with immunotherapy in patients with advanced non-small cell lung cancer (aNSCLC). The group developed a list of considerations for the design, conduct, and interpretation of RWD studies from different data sources.
rw-Response established a framework for measuring rw-response to treatment and found relative consistency of the measure across RWD sources in first-line chemotherapy regimens for patients with aNSCLC.
rw-Care developed a framework for the use of RWD at point of care to improve patient outcomes from immunotherapy by mitigating treatment-related adverse events.
Traditional clinical trials, such as randomized controlled trials (RCTs), can take a long time and typically enroll a narrow patient population that represents a fraction of the patients who will likely receive the product in the real world. RWD can generate supplemental evidence that reflects a larger and more diverse patient population than is included in clinical trials and address timely clinical questions including use of different treatments in standard of care practice and long-term safety. Without Friends coordination and support from collaborative sponsors, groups may never agree on a solution for aligning best practices and frameworks for aggregating and analyzing RWD.
Portfolio Timeline
Project Outcomes
2023
- A manuscript describing challenges and opportunities for aligning electronic patient reported outcomes data to generate RWE was published in JCOCCI.
- In June, Friends presented a poster about the rw-Response Findings at the ASCO Annual Meeting.
- In September, Friends hosted a public meeting titled Supporting the Use of RWD in Oncology Drug Development. The meeting presented results from the Friends real-world Response (rw-Response) Pilot. Click here to view the meeting presentation. A discussion document was also released, titled Considerations for Leveraging Real-World Endpoints in Oncology Drug Development.
2022
- Friends begins the rw-Response Project.
2021
- Results of the RWE Pilot 2.0 were published in Clinical Pharmacology and Therapeutics:The Friends of Cancer Research Real-World Data Collaboration Pilot 2.0: Methodological Recommendations from Oncology Case StudiesReal-world Overall Survival Using Oncology Electronic Health Record Data: Friends of Cancer Research Pilot
2020
- The 2020 white paper, Considerations for Use of Real-World Evidence in Oncology: Lessons Learned from Friends of Cancer Research Collaborations, identified the implications of dataset variability on measuring real-world endpoints and recommendations for developing a framework to guide future RWE studies through a harmonized protocol.
- Friends hosted a virtual event in September 21-22 An International Framework for Real-World Evidence, sharing the RWE Pilot 2.0 results and discussing potential policy and regulatory needs presented by this work.
2018
- Friends hosted a public meeting titled The Future Use of Real-World Evidence on July 10. The meeting white paper, Establishing a Framework to Evaluate Real-World Endpoints, details Pilot 1.0 and a framework for operationalizing and validating real-world evidence in advanced NSCLC.
Project Partners
Pilot 1.0: COTA, the U.S. Food and Drug Administration (FDA), Flatiron Health, IQVIA, Kaiser Permanente/Cancer Research Network (CRN), Mayo Clinic & OptumLabs, and National Patient Centered Cancer Research Network (PCORnet)/University of Iowa
Pilot 2.0: Aetion, American Society of Clinical Oncology (ASCO) CancerLinQ & ConcertAI, COTA, FDA, Flatiron Health, IQVIA, Kaiser Permanente, Mayo Clinic & OptumLabs, McKesson, National Cancer Institute (NCI) SEER-Medicare Linked Database, Syapse, Tempus AI Inc.
rw-Response Pilot: Ambra Health, ASCO, Columbia University, ConcertAI, COTA, FDA, Flatiron Health, Guardian Research Network, IQVIA, Ontada, Syapse, Tempus AI Inc.
rw-Care: Apricity Health, Carevive Systems Inc, Clinical Outcomes Solutions, Invitae, Navigating Cancer, Varian a Siemens Healthineers Company
